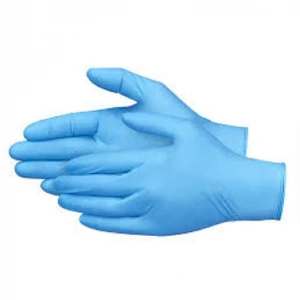
Disposable gloves are among the most commonly used protective
products. Bacteria, mycobacteria, fungi, microbes, and viruses
can easily move from touched objects onto your hands. Following
the basic rules of hand hygiene together with the use of
appropriately selected gloves protect the user, the patient,
and the touched object.
Examination and protective gloves are both a medical device and
a personal protection equipment. Their role is to protect hands
and to be the first obstacle to the spread of bacteria,
viruses, and other pollutants.
The quality of gloves and their characteristics are determined
by the restrictive and detailed guidelines described in the
relevant harmonized standards and directives.
Features od examination of protective gloves:
- evenly rolled cuff ensuring easy donning,
- ambidextrous,
- broad range of sizes: XS (5–6); S (6-–7); M (7–8); L (8–9); XL (9–10),
- they fulfil the requirements of Medical Device Directive 93/42/EEC and PPE Regulation (EU) 2016/425 (in accordance with Article 46 of Regulation (EU) 2016/425: Council Directive 89/686/EEC has expired with effect from 21 April 2018. References to the repealed Directive shall be construed as references to the Regulation in accordance with the correlation table in Annex X).
















 Browse Through Millions of High-Quality Products.
Browse Through Millions of High-Quality Products.